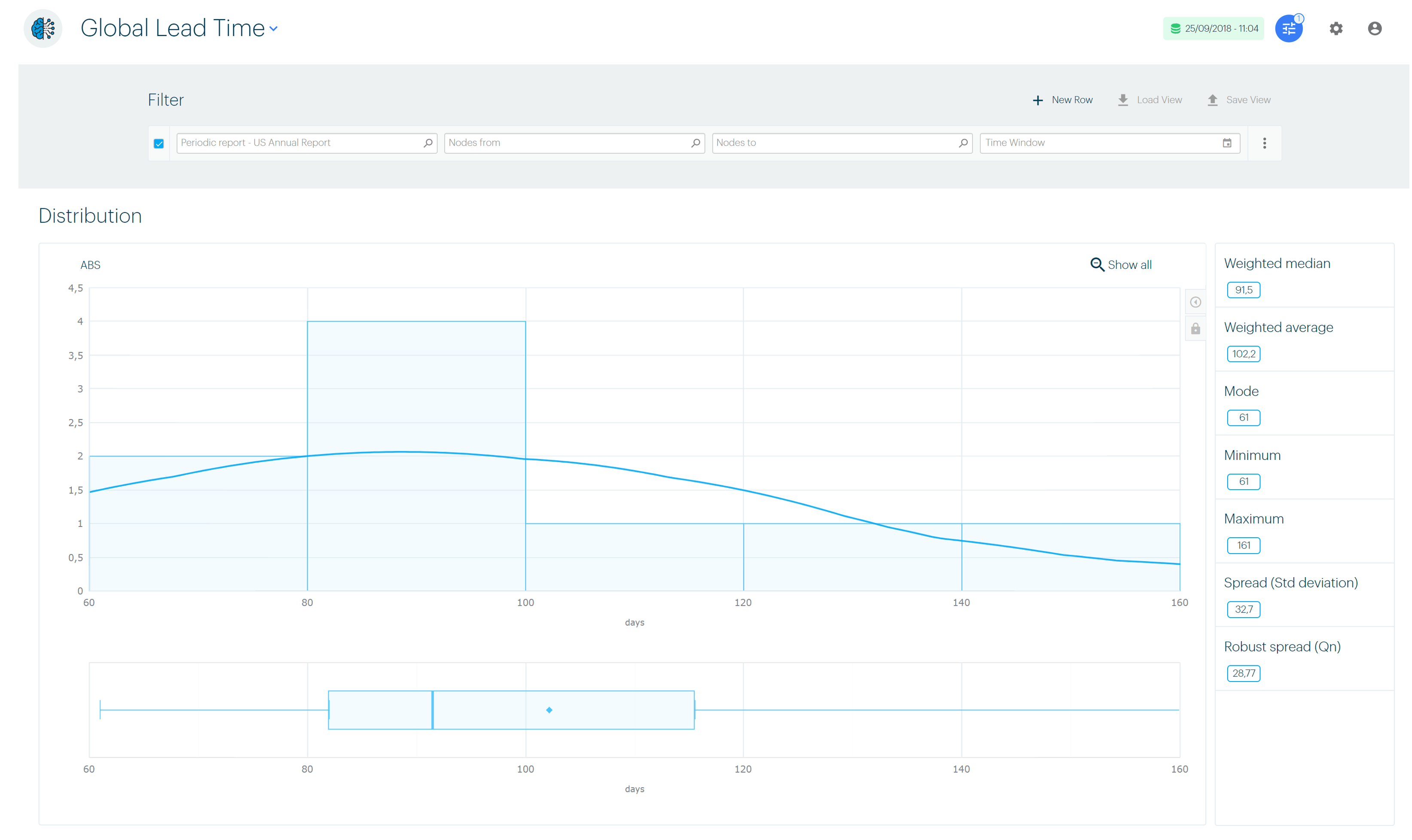
Everybody aligns around the same end-to-end standard lead times
Business context
We are a global regulatory affairs department that submits 1000+ submissions/year. We need to make and support delivery commitments to regulatory authorities and our internal supply chain and production partners. It is an understatement when we say that this is challenging.
Improvement potential
Before we implemented Binocs, delivery promising was ad hoc. It depended on who you asked. Some would confirm a technical variation submission in 6 months, other in 8 months. Moreover, we had a hard time delivering on our promise. Our planning adherence KPI was hardly 60% for some submission types.
How Binocs helped
We agreed on standardized work, which means that we defined “normal” lead times and workloads for the majority of our work item types such as “CMC Writing” and “Publishing”.
We also aligned around the “normal” submission process, which we configured in the Binocs Operating Model and which became the reference for our operation.
We went through some change management to get there, but these two aspects of standardization opened up a huge improvement in throughput and planning adherence.
But the wealth is often found in the simple things. We discovered how valuable it was that finally, our staff all made the same delivery promise. It creates trust and enables lean flow, not only in the GRA department but also with our partners in supply chain and R&D.
We also measured our lead times and using Binocs data science analytics we are now able to regularly tune our work standards to meet the reality. We are probably the only Regulatory Affairs department in the world that closed this loop using state-of-the-art data science.