How to become more efficient in cell and gene manufacturing — Resource planning
The Global Cell And Gene Therapy Market Is Estimated To Be Worth More Than £35Billion By 2026.
With the past year seeing Kite Therapeutics and Novartis make headlines for their groundbreaking therapeutic approvals, and a host of innovative biotech companies quickly advancing their production towards commercialization, the Cell and Gene industry is rapidly transforming the standard of care for rare diseases and critical conditions.
While there is a huge opportunity for industry growth, ever-present challenges prove barriers to achieving efficient commercial manufacturing scale.
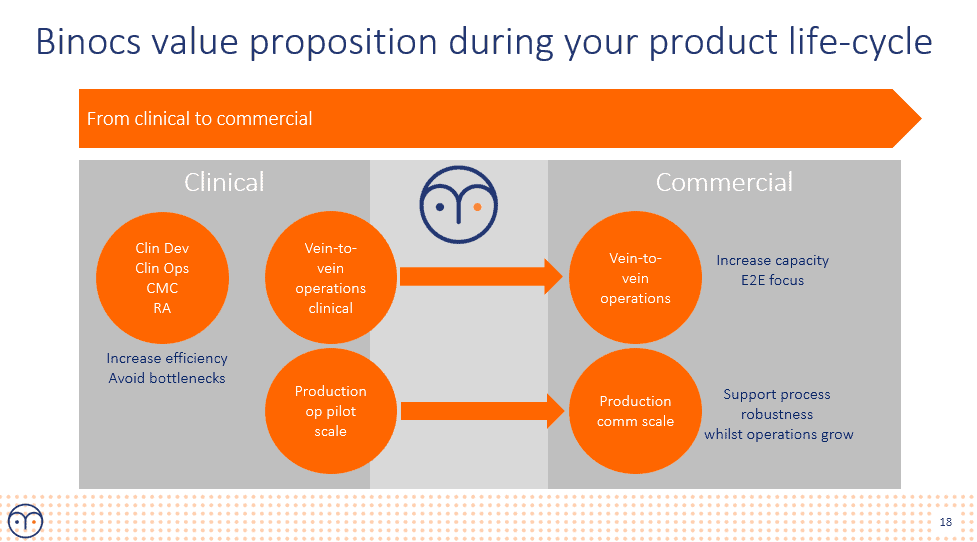
Efficient manufacturing resource planning, which is what?
Our colleague Joachim Lasoen attended the cell & gene therapy event last week (October 30th and 31st) with 2 goals in mind:
- Learn about the challenges and best practices in CGT manufacturing
- Present insights on the challenges in CGT resource planning and how Binocs can help solve them (based on experience at Novartis and Avexis)
Based on many conversations after the presentation it’s clear that, as many companies are reaching later stages in the development and commercialization is near, to become more efficient, manufacturing resource planning will become one of the cornerstones to drastically improve.
The very short processes with lots of rescheduling make traditional planning systems not suitable. Most companies thus end up with Excel files and lots of manual work. BUT it doesn’t have to be this way!
The Binocs manufacturing resource planning platform is specifically tailored to solve these challenges.